|
 |
| current issue |  |
past issues |  |
send a letter/news |  |
address update |  |
advertise |  |
about us |  |
alumni home |
Features
View FinderA professor's device may help doctors see where no MRI has gone before
By Robert Emro
 Nuclear physicist Bill Hersman in his UNH lab.
Nuclear physicist Bill Hersman in his UNH lab.
|
Magnetic resonance imaging can reveal the human body's inner workings in elegant detail, except in one place—the lungs.
But if UNH's second spin-out company is successful, MRI's blind spot may soon be corrected. Formed by Bill Hersman, a UNH nuclear physics professor, Xemed will try to capitalize on his innovations to give doctors an unprecedented look inside the lungs.
MRI scanners polarize the water molecules in our bodies, aligning the hydrogen nuclei with the poles of a powerful magnet. Responding to radio waves transmitted by the scanner, the polarized nuclei resonate with their own small radio waves, and a computer uses these signals to paint a clear picture of the interior of a human body.
Lungs, however, are filled with air instead of water, and show up as a hazy blur in conventional MRIs. One solution to the problem—giving a patient pre-polarized helium to inhale—produces sharp lung images. The drawback is that pre-polarized helium is prohibitively expensive. But xenon—present in tiny quantities in the air we breathe—is not. In addition, xenon has been used as an anesthetic in surgery and has a long track record of safe use in humans. It also dissolves into lung tissue and diffuses slowly, which makes it better at revealing damaged areas of the lungs. For all these reasons, researchers have been racing to polarize xenon.
One of the hurdles when using polarized gases in lung MRIs is that they are thousands of times less dense than water. To work, they must be highly polarized to levels thousands of times higher than normal. Researchers "hyperpolarize" helium by mixing it with vaporized rubidium, and exposing the mixture to a polarizing laser. Like a clutch transferring power from a car's engine to its wheels, the rubidium transfers polarization from the laser to the helium whenever the two collide.
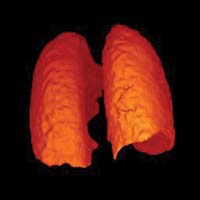 A computerized rendering of the volume of human lungs was created using hyperpolarized helium (3He).
A computerized rendering of the volume of human lungs was created using hyperpolarized helium (3He).
|
To increase the number of collisions between rubidium and helium atoms, the gaseous mixture is kept at high pressure so the atoms will be densely packed. Once cooled, the rubidium returns to its solid state, leaving behind pure hyperpolarized helium. Magnetic fields aid the polarization process and keep the gas in a polarized state until it is needed.
Adapting this process to xenon is an interesting problem with potentially big financial rewards. International research teams with strong financial backing have been working on it for some time in Germany, Japan and elsewhere in the United States.
Enter Hersman and his team of graduate and undergraduate researchers. As a nuclear scientist, Hersman had been polarizing gas for years to help shed light on the behavior of subatomic particles called quarks. With a small grant from the National Institutes of Health, he set out to solve the problem of lung MRIs.
Faced with a shoestring budget, Hersman brought a little Yankee ingenuity to bear. He knew that at low pressure, rubidium would combine with xenon to form short-lived molecules, a more efficient way to transfer polarization. To take advantage of that, however, he would have to come up with a way to effectively polarize rubidium at low pressure. His solution was amazingly simple. Instead of pointing the laser in the same direction as the gas flow, he turned it around and pointed it the other way. He also made the device's glass cylinder much longer to give the rubidium more exposure time. Hersman's methods produce xenon that is 60 to 70 percent polarized, better than any other method.
"Changing any one thing—lowering the pressure, increasing the flow or lengthening the cylinder—would make the system worse, but changing all three together resulted in polarization 10 times better than any other method," says Hersman. "And with a bigger laser, we can go up to a factor of 30 to 100 better."
Page: 1 2 Next >
Easy to print version
blog comments powered by Disqus